FST-344
Follistatin
Minicircle’s Follistatin gene therapy (FST-344) is a well-tolerated longevity plasmid gene therapy that can increase lean mass and decrease fat.
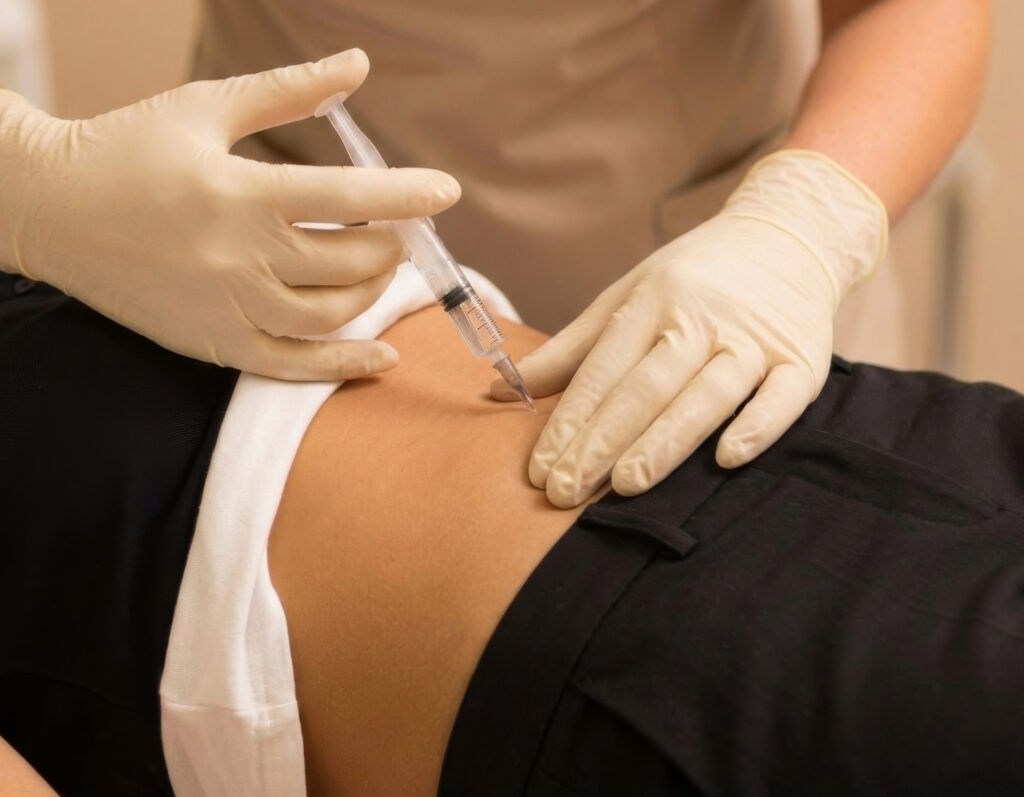
- Administered via subcutaneous injection
- Non-heritable effects that last ~1 year
- Now available at select clinics
Follistatin gene therapy uses DNA plasmids to temporarily deliver the follistatin gene to the cell for secretion, subsequently increasing whole-body follistatin protein. Follistatin blocks myostatin and activin. Myostatin is a protein that limits muscle development while activin plays a role in inflammation. Follistatin’s ability to improve body composition is dependent on both myostatin and activin inactivation.
Because it does not integrate with your chromosome, Follistatin gene therapy can boost Follistatin production without permanent genetic changes. Further, our clinical trial participants experienced no severe adverse effects.
Minicircle’s Follistatin gene therapy is administered via subcutaneous fat injection.
Minicircle’s Follistatin gene therapy is temporary as it does not integrate with your chromosomes. Treatment effects typically last for approximately one year, though individual experiences may vary.
Minicircle’s Follistatin gene therapy is well tolerated. The most common side effect observed is a slight increase (8mg/dl) in LDL cholesterol in about a third of patients, which can be managed through diet and lifestyle. We do not recommend Follistatin gene therapy for people who are pregnant or planning to become pregnant in the next year.
*Individual experiences and results may vary. Consult your primary care physician before receiving gene therapy.
Anticipated benefits of Follistatin gene therapy.
How it works.
Start your journey with Minicircle’s plasmid gene therapy today.
Contact a clinic.
Follistatin is currently available at state-of-the-art facilities in beautiful destinations across Central America. Get started here — we’ll help you set up a profile and find an appointment.
Complete testing.
A clinician will work with you to complete necessary testing and answer any questions you have. This ensures the best possible experience with novel gene therapy.
Receive therapy.
Travel to your clinic, where the therapy is administered via subcutaneous injection. We’re here to help you have a smooth experience, from travel logistics to treatment and aftercare.
Witness your transformation.
Track your results and hear about other therapies we’re launching through your personal portal and intuitive dashboard. We’re there every step of the way.
Move forward with Follistatin.
Plasmid gene therapy to help you pursue your purpose.
FST-344 therapy outcomes at 3
months.
Serious adverse events
bodyfat % change
lean mass increase
Epigenetic age reduction in
older patients*
*Measured via TruDiagnostic
What to expect.
Clients typically feel the physical effects of Minicircle gene therapy for approx. 12 months. It’s most effective when paired with a healthy, active lifestyle.
*Dates based on estimates. Individual experiences and results may vary.
Onset (30 days)
Begin to
feel effects
Boost (1-3 months)
See incremental
improvements
Maintain (3-11 months)
Consistent effects
Onset (30 days)
Begin to feel effects
Boost (1-3 months)
See incremental improvements
Maintain (3-11 months)
Consistent effects
The future of gene therapy.
We’re elevating human potential with an exponentially scalable gene therapy platform.

Get Follistatin at state-of-the-art clinics in Central America.
FAQs
Plasmid gene therapy delivers genetic instructions to your cells, enabling them to produce therapeutic proteins internally. Rather than frequent injections of an externally manufactured protein, Minicircle plasmids (circular DNA molecules) prompt the body’s own cells to synthesize the proteins. While results may vary, a single injection of plasmid can support protein production for an entire year, depending on the individual’s unique biochemistry. Learn more.
Minicircle’s therapy uses a single subcutaneous injection to deliver the therapeutic plasmid.
- The plasmid enters the nucleus but does not integrate into the genome.
- The cell produces the encoded protein (e.g. follistatin (FST)), which circulates in the bloodstream.
- The therapeutic effect depends on the specific protein and the individual’s response. For example, FST inhibits myostatin and activin in order to increase lean mass.
Follistatin inhibits myostatin and activin, proteins that suppress muscle growth. By reducing these inhibitors, FST may help support increasing lean muscle mass, decreasing fat mass, and reducing epigenetic age.
Minicircle gene therapies are administered as a single subcutaneous injection by licensed physicians in clinical settings.
In over 500 patients treated to date, Minicircle’s FST gene therapy has been well-tolerated. The most common reported side effect has been a slight increase in LDL cholesterol (8mg/dl) in roughly one-third of patients. This increase is not clinically significant and can be managed through diet and lifestyle changes.
No. FST gene therapy is not recommended for individuals who are pregnant or planning to become pregnant within 12 months of treatment.
While effects vary, patients receiving FST therapy report increased lean body mass, decreased fat mass, and improved strength and physical performance.
Recommended assessments include DEXA scans to track body composition changes, as well as performance tracking and physician evaluations. Epigenetic age tests are currently under review. No specific test is officially recommended at this time.
Participants must be 23 years or older based on Institutional Review Board (IRB) guidelines.
Minicircle is preparing clinical trial data for publication. Please subscribe to our newsletter or follow us on social media for updates as data becomes available.